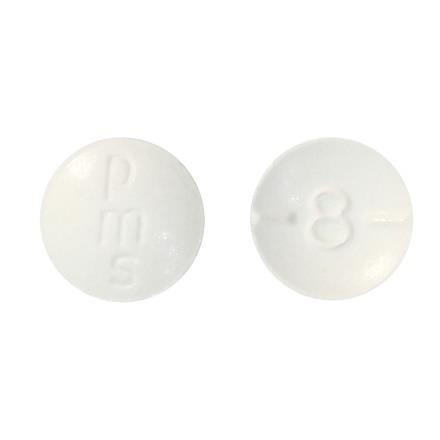
Recall of one lot of pms-Hydromorphone due to packaging error that could potentially lead to overdoseConsumer ProductsAug 20th, 2022 @ 12:00 PM
Basic Details
Category: Health product - Drugs
Alert type: Public advisory
Issue: Health products - Adverse reactionHealth products - DosageHealth products - LabellingHealth products - PackagingHealth products - Product safetyHealth products - Product withdrawal
Product: pms-Hydromorphone, 2 mg tablets (DIN 00885436) marketed by Pharmascience Inc. Sold to pharmacies in bottles of 100 tablets for dispensing to consumers.
What to do: Check your bottle of pms-Hydromorphone to ensure you have the correct tablets. Stop using product from the affected lot and contact your pharmacy immediately for a replacement product.
ImagesClick to enlarge
Products
| Lot | Expiry date | DIN | Product |
|---|---|---|---|
| 639268 | 31-01-2026 | 00885436 | Pharmascience Inc. pms-Hydromorphone 2 mg Hydromorphone Hydrochloride Tablets Bottles of 100 tablets |
 Recall Monitor
Recall Monitor
Information presented from Recalls and Safety Alerts dataset, provided by the Government of Canada through the Open Government initiative and is subject to the Open Government License.
You can access this data free of charge via Recall Monitor, or through the Healthy Canadians website.
Get personalized SMS or Email notifications about recall alerts that matter by subscribing to Recall Monitor.
Recall Monitor is not endorsed by, or is an official product of the Government of Canada.
Made with in Vancouver, BC
© Mad Filaments Software Inc.